This is the second abstract that will be presented as poster at AACR in June. If you are interested in learning more about the project or how zebrafish models could be used as a tool for precision medicine, contact us at info@bioreperia.com or in the contact form below.
Titel: Translation of zebrafish tumor-derived xenograft-models for improved diagnosis and treatment planning in urinary bladder cancer patients
Authors: Zaheer Ali, Anna Nilsson, Malin Vildevall, Ylva Huge, Amir Sherif, Anna Fahlgren, Lasse DE Jensen
Introduction
Precision medicine in oncology aims to identify the most effective treatment for any given patient based on individualized analyses of patient material. Currently, precision medicine relies on sequencing of DNA or RNA to identify patient tumor-specific mutational profiles that may be coupled to drug response. These techniques, however, fail to reveal actionable mutations in approximately 85% of the cancer patients, and have not been established at all for many commonly used drugs including cisplatin-based treatments used in urinary bladder cancer. While mouse-PDX models can determine drug response rates with high accuracy in most patients and for most drugs, such techniques are too slow and expensive to be relevant for first line treatment planning. Urinary bladder cancer patients are often treated with cisplatin-containing combination therapy, with the hope of down-staging tumors before surgery. 60%, however, do not respond or even progress on this treatment, and these patients would benefit from immediate surgery upon diagnosis. To help identify non-responding patients, we show here that patient-derived tumor xenograft models can be established in zebrafish larvae (ZTX models) and that the resulting tumors exhibit differential responses to the two main cisplatin containing treatments GC and MVAC.
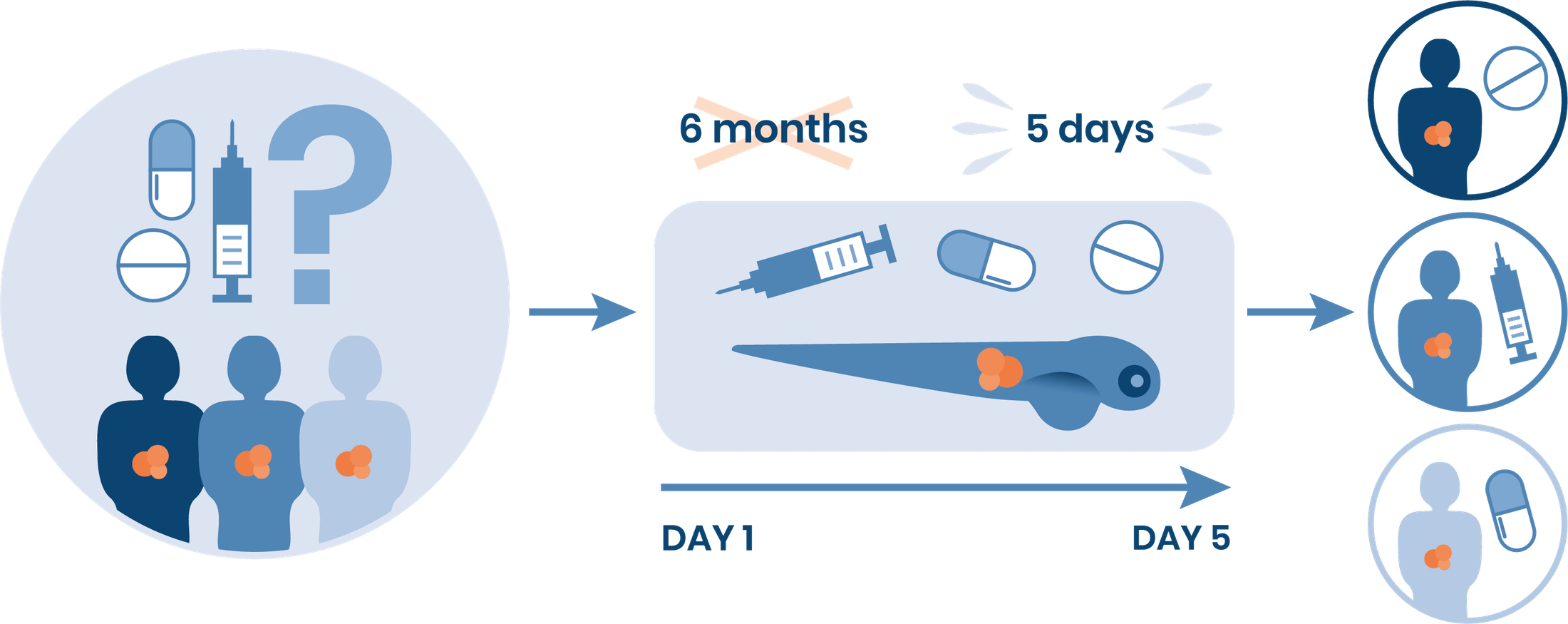
Results
Preliminary results from the first 19 patients are presented. Two tumor biopsies were destroyed during transport and two did not allow isolation of sufficient viable cells for implantation. From the remaining 15 samples an average of 2,6 million cells with average viability of 53% were isolated and used to implant at least 60 2-days old larvae. All 15 samples implanted in the larvae and survived and/or grew exhibiting varying degrees of metastatic dissemination (average between 2 and 13 metastasized cells per embryo and model) within only three days from implantation. Four ZTX models exhibited different responses to GC and MVAC demonstrating that these treatments are not equally effective in all patients. Non-response in ZTX models was associated with tumors having re-appeared in the bladder upon radical cystectomy in all patients undergoing surgery prior to Dec. 5th 2019 (n=3). GC inhibited metastasis in all models (average 69% inhibition), whereas MVAC inhibited metastasis in 40% of the models (average 36% inhibition).
Since the abstract was submitted, new data has been provided, showing correlation between the ZTX model and the clinical outcome in patients at 100% (n=5).
Conclusion
The ZTX urinary bladder cancer platform presented here overcome limitations associated with long assay time and high cost of other functional models within precision medicine as well as the low hit-rate of actionable mutations associated with genomic techniques. ZTX models will therefore likely become a powerful method for functional precision medicine within oncology, in the near future.
The poster will be presented in session PO.TB01.06- Model Organisms for Cancer Research and have the code 6124/8

